  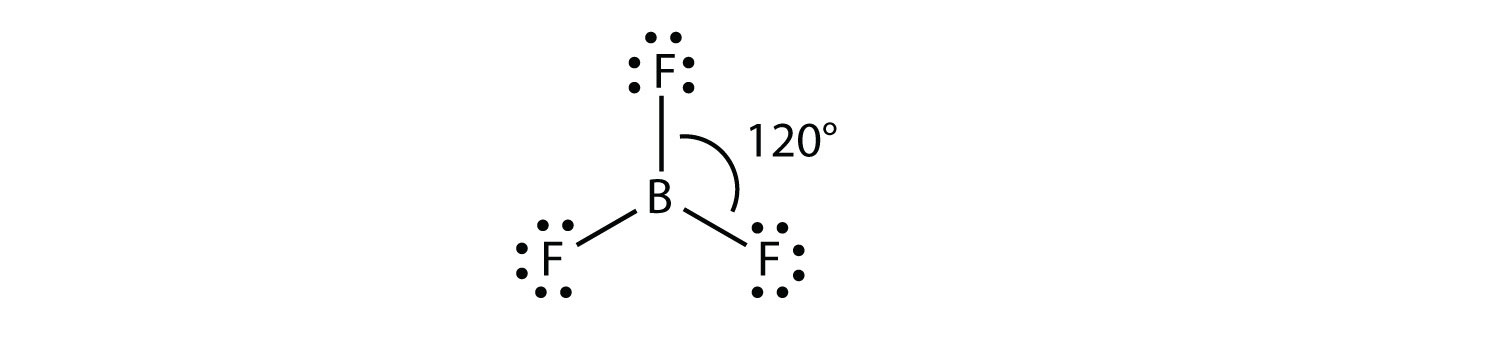
Electron Pair Geometries Unear Trigonal planar Tetrahedral Tetrahedral. electron pair geometry of pf3 What Is The Electron Geometry Of Pf3 -. An explanation of the molecular geometry for the SCl2 (Sulfur dichloride) including a description of the SCl2 bond angles. Transcribed image text: Draw the Lewis structure for PF3 (phosphorous is the central atom) and select the correct model representing its 'electron pair geometry and molecular geometry'. This is because electrons distribute around an atom to minimize repulsion with one another. By knowing the electron domain of each atom in a molecule, you can predict its geometry. 
It represents the number of locations expected to contain electrons. Trending Questions Where can I get free math lesson online? What are the answers to don't know much about liberty? What are the x and y coordinates for Milwaukee? What numbers have 3 factor pairs that are less than 20? Who discovered the formula for the area of the parallelogram? What is Sam known for? Why do you need tolerances on dimensions? Sum and differences of rational algebraic expressions? Fahrenheit 451 is written in the third person he What Is mean? Why do you multiply and divide functions? Why is 51 a composite number? The caboose is 9. The HBeH H Be H bond angle is 180o 180 o because of its linear geometry. domain geometry for a molecule with four electron pairs is tetrahedral. An atom's electron domain is the number of lone pairs or chemical bond locations that surround it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
